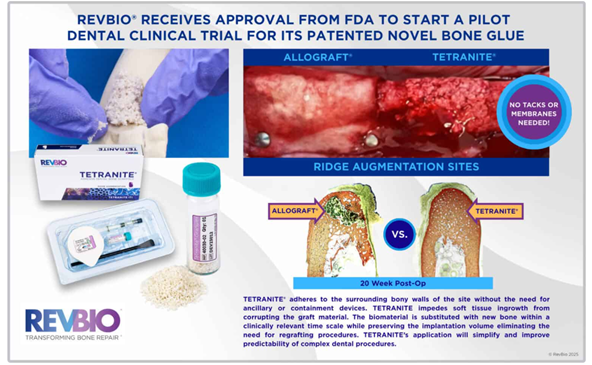
A Breakthrough Bone Adhesive for Dental Procedures
December 22, 2025 – Boston, USA – RevBio, Inc., a clinical-stage medical device company, has received U.S. FDA approval on 22nd Dec 2025 to begin a pilot clinical trial for TETRANITE®, a regenerative bone adhesive designed for dental ridge augmentation. This innovative biomaterial allows dentists to perform bone grafting procedures without membranes, meshes, or fixation aids such as tacks or screws, streamlining complex dental surgeries and potentially improving outcomes.
How TETRANITE® Works
- TETRANITE® is engineered to adhere directly to the surrounding bone, preventing soft tissue from interfering with the graft.
- Over time, the adhesive is naturally replaced by new bone while maintaining the graft’s volume, reducing the need for additional grafting procedures.
- The product is intended to simplify dental ridge augmentation, particularly for patients with missing teeth and significant bone loss, where traditional grafting materials may fail in up to 30% of cases.
Clinical and Research Support
The development of TETRANITE® was supported by $1.8 million in grants from the Translational Resource Center (TRC) under the National Institute of Dental and Craniofacial Research (NIDCR), along with a $2 million SBIR Phase II grant. These funds enabled preclinical research and preparation for human trials.
Dr. Rahul Jadia, RevBio’s R&D Manager, stated, “TETRANITE® adheres to bone and supports new bone growth without losing volume or strength, offering reliable results for complex dental procedures.”
Advantages for Dental Professionals
- Eliminates the need for secondary fixation or containment devices
- Reduces procedural complexity and operating time
- Supports predictable bone regeneration, improving implant readiness
- Potentially decreases the need for repeat grafting and overall treatment costs
Bioengineered for broad applications in dental surgery, including ridge augmentation
About RevBio, Inc.
RevBio is a Boston-based clinical-stage medical device company focused on regenerative technologies for dental, orthopedic, and neurosurgical applications. TETRANITE® is currently in multiple FDA-approved human trials and is anticipated to receive De Novo classification for key indications, accelerating its path to commercialization.
Looking Ahead
TETRANITE® represents a promising advance in dental biomaterials and regenerative medicine. Its upcoming clinical trials will evaluate safety and effectiveness in humans, with the goal of improving outcomes in dental implant therapy and other complex procedures.
For dental professionals, this technology may provide a more efficient, reliable, and predictable approach to bone grafting, highlighting the growing impact of bioengineered solutions in modern dentistry.
REFERENCE
- RevBio® Receives FDA Approval to Start Dental Clinical Trial
Business Wire / Yahoo Finance — Announcement of FDA approval to begin the dental clinical trial for TETRANITE®. (Yahoo Finance)
https://finance.yahoo.com/news/revbio-receives-approval-fda-start-181200269.html - Official Business Wire Press Release — RevBio FDA Approval
Business Wire — Press release detailing FDA approval for the pilot clinical trial of the regenerative bone adhesive. (Business Wire)
https://www.businesswire.com/news/home/20251219800454/en/RevBio-Receives-Approval-from-FDA-to-Start-a-Dental-Clinical-Trial-for-its-Regenerative-Bone-Glue